Exploring the IR and Raman signatures of substituted biological apatite – evaluating the effect of orientation on specific spectral bands
Organization: Department of Physics – Josip Juraj Strossmayer University of Osijek, Universität des Saarlandes, Physikalische und Theoretische Chemie
Discipline: Matter – knowledge based multifunctional materials
Objectives
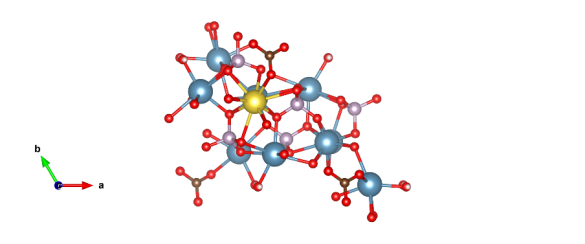
Apatites presents a group of compounds with a similar atomic structure. The first step before actual knowledge of the exact structure was acquired in 1930, where X-ray pattern discovered apatite in tooth and bone mineral. Biological apatite is an inorganic, crystalline calcium phosphate material with an atomic structure similar to the abiotic hydroxylapatite (OHAp) that forms the main component of biological hard tissues, such as bone, dentin, and enamel. Chemical formula of the apatite is X10(YO4)6Z2 where X, Y and Z can be substituted with different elements. Depending on the elements substitution, distinction between varius apatites can be made. For example, some of the substitutions which can be made is X=Ca, Ba, Sr, Pb, etc., Y=P, Mn, Mg, Na, Li, etc., and Z=OH, C, Cl, Br, etc.
In this work, attention is on the hydroxylapatite with Ca, OH, P, Na and C substitutions. Due to this, significant amounts of research have been undertaken in the materials science and medical science domains as changes in the chemical reactivity of the material can be related to pathological mineralisation, such as in cardiovascular disease deficient mineralisation, including osteoporosis, and are important for the biocompatibility of bone and teeth implants. Using IR and Raman spectroscopy techniques, both in theory and experiment one can easily obtain information about chemical properties for each molecular group in the particular mineral.
In this project we aim to answer the following research questions using computational simulations:
- What is the extent of orientation dependence on the relative intensities of the carbonate and phosphate bands in vibrational spectroscopy data?
- Do measured differences originate entirely from the applied incoming light source direction or does preferred growth morphology and nanometre size of the crystals, i.e. dominance of surface over bulk phonon signatures, also result in spectral changes?
Achievements
In the beginning of our work we identified the correct structures, defined appropriate basis sets, obtain relaxed structural parameters and geometry. After that, we calculated IR and Raman spectra and done vibrational mode analysis of active modes. Next task was to cut bulk structure to polar and non-polar low Miller index surfaces of OH-apatite in correspondence to available literature and previous works and to compute electronic ground state geometry of 2D relaxed slabs. Our approach of obtaining slabs is different from one we found in literature in terms of assessing the dipole moment during the cleaving process. We select our surfaces in the way that tetrahedral species maintain their geometry, which significantly reduces the number of possible surfaces in the otherwise vast combinatorial space. For slabs, IR and Raman spectra was investigated and evolution of mode with respect to increasing number of layers which is linked to the experiments. With substitutions we obtain three types of hydroxylapatites, type A, type B and type AB.
This work and collaboration will continue after the project is officially completed, which is the greatest achievement of all.
What a scientific collaboration actually looks like
This project gave me the opportunity to see what a scientific collaboration actually looks like and what it takes to make it a success. I realized that progress of science requires collaboration between scientist and researchers no matter where they are at the moment. Every discussion enriches the individual, especially when it takes place intensively over a certain period of time, as it was in my case. The combination of young, enthusiastic scientists and those more experienced represents a winning combination for prosperity. It is important to note that this project didn't end with my departure, but is still ongoing. This project has allowed me to expand my contacts and some of them are already starting to manifest themselves through future scientific collaborations.
More information on HPC-Europa3: www.hpc-europa.eu

0 Praat mee